The Science of Dopamine Restoration Through Regenerative Therapy
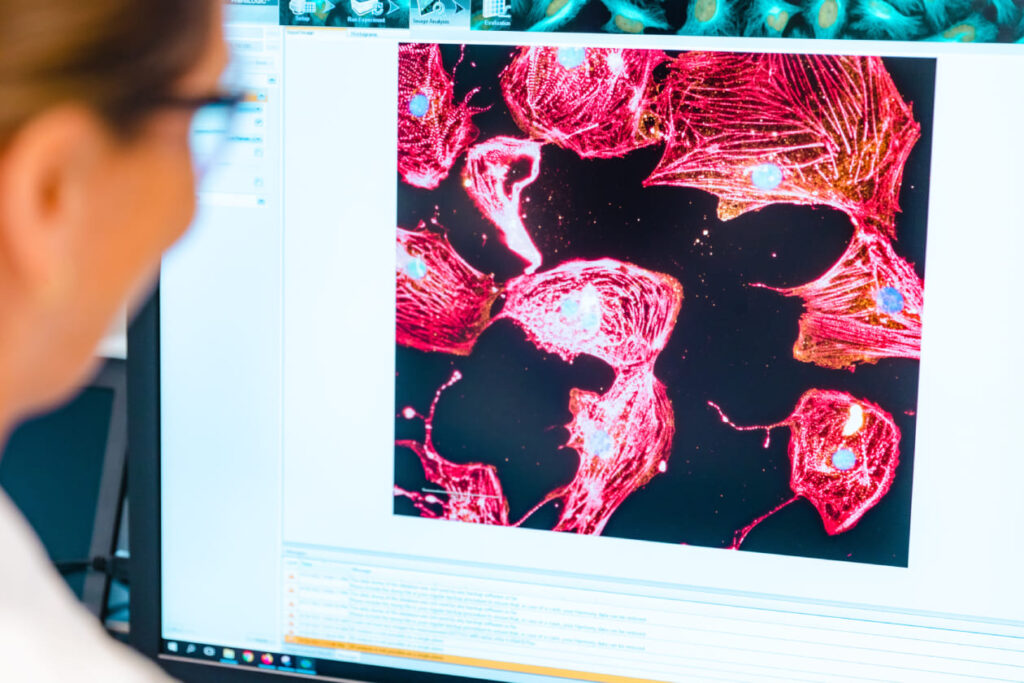
Dopamine supports movement, focus, and motivation by enabling communication between nerve cells in the brain. Changes in dopamine activity are linked with several neurological conditions that affect daily function and quality of life. Regenerative therapy is being explored in clinical research as a way to support the biological environment connected to these pathways. In regenerative…