
by admin | Oct 30, 2018 | Studies, Stem Cell Research, Stem Cell Therapy
Spinal cord injury is the second leading cause of paralysis in the United States. When the spinal cord is severely injured, nerve cells in the spinal cord are damaged or destroyed. Also, a sort of scar forms in the affected area, which prevents nerve signals from traveling between the brain and the extremities. Consequently, people who sustain spinal cord injuries suffer from paralysis. The degree of paralysis depends on the location of the spinal cord injury; injuries higher on the spinal cord such as the neck or upper back area can lead to paralysis of all four limbs, for example. In almost all cases, the paralysis is permanent once it occurs, because nerve cells in the spinal cord do not regenerate.
Because spinal cord injuries are common and the consequences are usually permanent, researchers have been aggressively and tirelessly researching ways to treat this condition. One approach is to try to form new nerve cells in the spinal cord using stem cells. Mesenchymal stem cells can become new nerve cells given the right set of circumstances. Unfortunately, simply injecting mesenchymal stem cells into patients with severe spinal cord injuries cannot reverse paralysis. On the other hand, using exosomes from mesenchymal stem cells may be the push that stem cells need to become nerve cells in the spinal cord.
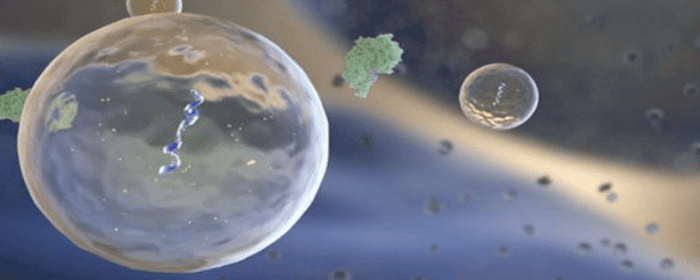
Exosomes are tiny packets of cellular material released by stem cells. They contain a variety of potentially beneficial substances; perhaps the most important in cell regeneration is micro RNA (miRNA). miRNA can cause complex changes in cells that simple drugs, proteins, or even regular RNA cannot. Researchers cannot easily deliver miRNA to where it is needed in the body, but exosomes taken from stem cells can deliver miRNA right where it needs to be.
Researchers collected human mesenchymal stem cells and placed them in an environment that would cause them to become nerve cells. But instead of simply using the stem cells directly, they instead collected the exosomes from those stem cells. Those exosomes could then be used to prompt mesenchymal stem cells to become nerve cells. Simply put, the exosomes drove the process more efficiently than the stem cells alone.
What does this all mean? Exosomes taken from the mesenchymal stem cells could eventually be used to treat spinal cord injury. Those special exosomes would magnify the nerve cell-creating effect, perhaps restoring nerve cell function to a damaged spinal cord. Considerable research needs to be done before this possibility becomes a clinical reality, but this knowledge helps researchers design targeted experiments in the future.

by admin | Oct 11, 2018 | Stem Cell Research
Myocardial infarction, also known as heart attack, can be a devastating or even deadly event. It occurs when blood flow in one or more coronary arteries is blocked. Since coronary arteries supply blood to the heart muscle, a blockage in a coronary artery prevents oxygen and nutrients from reaching heart tissue.
While the heart can sustain short periods of time without oxygen or nutrients, heart cells become dysfunctional and die if blood flow is not restored within several hours. While clot-busting drugs, percutaneous intervention (PCI), and balloon angioplasty have provided a way to restore blood flow to the heart during a heart attack, once heart cells die there is no way to bring them back. Since most heart tissue is cardiac muscle, dead heart tissue cannot participate in the contraction or squeezing of the heart during a heartbeat. Thus, people who have survived a heart attack are often left with poor heart function (e.g. congestive heart failure).
Stem cell researchers have begun to question whether heart tissue destroyed during a heart attack is necessarily gone forever. Research is beginning to show that stem cells given after myocardial infarction are able to improve the squeezing power of the heart. By extension, stem cell treatment is able to improve the abilities heart to pump blood throughout the body.
Researchers initially assumed that it was the stem cells themselves that became new heart cells, replacing dead and dysfunctional heart tissue. While there is evidence that this occurs, it seems that stem cells play an even bigger role in heart tissue repair than simply becoming new heart cells. Stem cells release small packets of a material called exosomes and microvesicles. Exosomes and microvesicles hold proteins, cytokines, chemokines, growth factors, DNA, messenger RNA, and micro RNA. Researchers now believe that these materials hold the true power of stem cells in cardiac repair and regeneration.
Various types of stem cells produce exosomes that could potentially help repair a damaged heart. While cardiac stem cells may seem like an obvious source for these exosomes, induced pluripotent stem cells and mesenchymal stem cells are also capable of releasing exosomes that are potentially beneficial in cardiac repair.
Stem cells—or more accurately the exosomes contained within the stem cells—help repair damaged heart tissue in several ways. Stem cell-derived exosomes contain factors that promote the survival of vulnerable heart cells and cells that are dysfunctional after a heart attack (but not dead). Exosomes also help new blood vessels to form in and around the damaged heart muscle in a process called angiogenesis. These new blood vessels deliver oxygen, nutrients, and molecules that help support the growth and function of heart tissue. Exosomes also appear to promote a healthy immune system response after a heart attack, rather than a destructive inflammatory reaction. In other words, the materials found in exosomes guide the immune system to clear away damaged tissue without creating extensive fibrotic (i.e., tough, nonfunctional) tissue.
While most clinical trials thus far have studied the effects of stem cells directly infused into humans after myocardial infarction, exosomes are rapidly becoming the focus of future clinical trials in this area.

by admin | Aug 14, 2018 | Bone Marrow, Stem Cell Research
Colitis is inflammation of the colon, also known as the large intestine. Several things can cause colitis such as infection, medication, ischemia, or chronic inflammatory bowel disease. Inflammatory bowel diseases that affect the colon, such as ulcerative colitis or Crohn’s disease, are particularly challenging for patients. It is a chronic disease that causes cramping pain, bloody diarrhea, weight loss, fatigue, and many other chronic, challenging symptoms.
Since ulcerative colitis does not occur naturally in animals, researchers sometimes use an experimental form of colitis to mimic the disease seen in humans. This experimental colitis serves as a model to investigate treatments for inflammatory bowel disease. Essentially, researchers create a situation in which mice develop a condition that looks very much like ulcerative colitis. They develop inflammation in the large intestine, along with signs of oxidative stress and cell death. Conversely, treatments for ulcerative colitis reduce or prevent inflammation, oxidative stress, and cell death in the colon of these experimental mice.
Researchers used this model of experimental colitis to study the effect of bone marrow stem cells as a treatment for colitis. More specifically, they tested the effects of a certain part of bone marrow stem cells called extracellular vesicles. Extracellular vesicles are small spheres that containing various beneficial substances. Stem cells release these vesicles into the body. A single stem cell can release hundreds of extracellular vesicles. In fact, it is the extracellular vesicles that are believed to contain many of the useful substances that are released by bone marrow stem cells such as proteins, lipids, and nucleic acids. These substances can precisely target sick and damaged cells in the body and repair them.
Impressively, when researchers used extracellular vesicles derived from bone marrow stem cells to treat animals with experimental colitis, they observed rather extraordinary results. These vesicles protected the intestines from colitis damage. Untreated animals had severely damaged intestines when viewed under a microscope, but animals treated with extracellular vesicles had nearly normal looking intestines. Treatment also substantially reduced levels of cytokines related to oxidative stress, such as IL-1β. Extracellular vesicles derived from bone marrow stem cells also apparently blocked the intestinal cells’ ability to undergo cell suicide (apoptosis).
Taken together, these results strongly suggest that mesenchymal stem cells from bone marrow, specifically the extracellular vesicles contained within them, can dramatically improve experimental colitis. While more research is needed, this study suggests that these stem cell products could one day be a useful treatment for inflammatory bowel diseases, such as ulcerative colitis and inflammatory bowel disease.


 St. Petersburg, Florida
St. Petersburg, Florida
