
by admin | Jan 16, 2017 | Studies
In recent years, there have been a number of advances in stem cell research and in the various ways that these cells can be best employed to improve the health of patients. One of the medical areas that shows promise for stem cell therapies is orthopedics, with stem cell therapies being developed to help with tissue, cartilage, and bone repair. A recent review by Anish Majumdar and colleagues conveyed the progress that has been made specifically in the use of stem cells for the repair of cartilage in osteoarthritis.
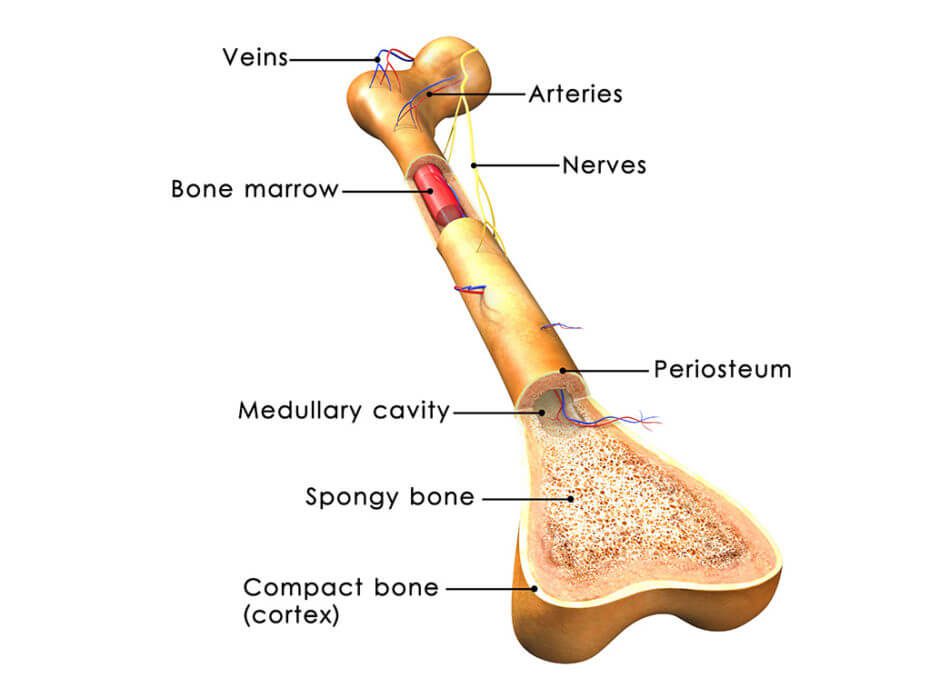
Patients with osteoarthritis experience degeneration of their connective tissues, which progresses as they get older. While osteoarthritis is often diagnosed in older patients, athletes also often endure osteoarthritis after injuring themselves while playing sports. In osteoarthritis, tissue known as articular cartilage is particularly susceptible to injury and unfortunately does not heal as well as other tissues because it does not have the vasculature required to supply the tissue with the nutrients required for significant growth and recovery.
When articular cartilage is damaged, surgery is often employed in an attempt to correct the damage, and pharmaceuticals are sometimes prescribed for discomfort. However, these interventions do not tend to achieve satisfying results. Because bone marrow stromal cells, or bone marrow-derived mesenchymal stem cells, naturally differentiate into the cells that make up cartilage, it has been suggested that these stem cells could improve outcomes for those with osteoarthritis.
These particular stem cells have other advantages, including that their isolation is relatively simple and that they easily proliferate, adds to their attractiveness as a candidate for cartilage repair. Their ability to suppress the immune system and prevent inflammation makes them more likely than many other cell types to be safe when added to the cartilage. As such, according to this review, a number of researchers have reported that their injections of these stem cells in patients with osteoarthritis have not caused any problems related to safety.
These findings include stem cells leading to improvements in clinical symptoms and quality of life in those with osteoarthritis, as well as the filling of the defect area and reduction in pain.
Researchers have also reported that these stem cells are effective from a therapeutic standpoint when administered to osteoarthritis patients. A number of specific findings on the success of bone marrow-derived mesenchymal stem cells in cartilage repair demonstrate that stem cells could revolutionize therapeutic strategies for this type of tissue damage. These findings include stem cells leading to improvements in clinical symptoms and quality of life in those with osteoarthritis, as well as the filling of the defect area and reduction in pain.
See why more and more athletes are turning to stem cell therapy here.
Reference
Gupta, P.K., Das, A.K., Chullikana, A., & Majumdar, A.S. (2012). Mesenchymal stem cells for cartilage repair in osteoarthritis. Stem Cell Research & Therapy, 3(25), 1-9.

by admin | Nov 7, 2016 | Studies
A group of Japanese researchers have just released a study that advances our understanding of how we can use a specific type of stem cells to help patients recover from cartilage damage. This group worked with a specific type of stem cell called bone marrow-derived mesenchymal stem cells (BMSCs) because these cells confer a number of advantages over other types of stem cells that have been used to repair cartilage. Though one particular type of stem cells, called autologous chondrocytes, have been implanted over 20,000 times for therapeutic reasons, these cells are associated with long and arduous surgeries because it is difficult to harvest the required cartilage and periosteum. Another type of stem cell, referred to as suspended cultured chondrocytes, on the other hand, have the potential to result in leakage, with an uneven distribution of cells throughout the site.
Unlike these other stem cell options, BMSCs can be easily harvest through methods that are relatively minimally invasive. The team in Japan also believes that BMSC implantation is safe, as they observed no tumors or infections in the more than 6-year follow-up period of their study. Nonetheless, previous work with BMSC showed that the cells did not lead to the generation of cartilage that was sufficient to replace old cartilage. To overcome that problem, these scientists decided to explore ways to provide large numbers of cells over a short period of time. Because BMSC from humans lose their ability to differentiate into different types of cells after having traveled long distances, the researchers believed that injecting cells directly into the injury site would improve the chances that good cartilage would develop. In addition, adding a specific agent, called, fibroblast growth factor (FGF-2) could help the cells proliferate.
In this study, the researchers tested these ideas to help determine the ideal conditions for transplanting BMSCs for cartilage repair. They found that their method of transplantation led to better cartilage development than was seen in controls, with rats receiving BMSC treated with FGF-2 directly into the area with cartilage injury having higher Wakitani scores. Wakitani scores are used to assess the regeneration of cartilage tissue and how well the new tissue is integrated into the surrounding tissue.
Given that many cartilage defects do not tend to repair themselves and can often lead to osteoarthritis, it is important to find effective ways to regenerate cartilage. There are a number of limitations to the current therapies, but this study demonstrates how our tools for cartilage repair are progressing and the promise of stem cells to help rebuild defective cartilage.
Learn more about using stem cells for orthopedics here.
Reference
Itokazu et al. (2016). Transplantation of scaffold-free cartilage-like cell-sheets made from human bone marrow mesenchymal stem cells for cartilage repair. Cartilage, 7(4), 361-372.


 St. Petersburg, Florida
St. Petersburg, Florida
