
by admin | Jun 26, 2018 | Stem Cell Research
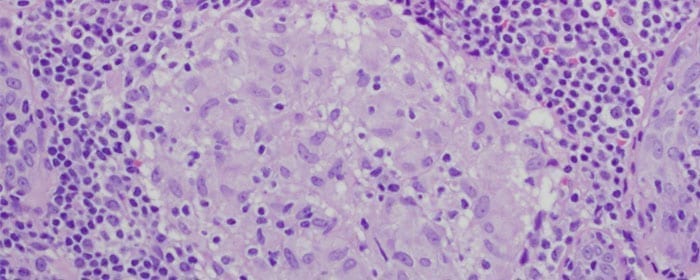
Sarcoidosis is a condition in which granulomas (non-cancerous masses) form in the body. While sarcoidosis can affect any organ, it most often affects the lungs and chest lymph nodes. The cause of sarcoidosis is unknown, but it is thought to relate to problems with the immune system. When sarcoidosis affects the lungs, patients often have coughing and wheezing, shortness of breath, and chest pain. In general, patients with sarcoidosis have a mild fever, joint swelling and pain, and extreme fatigue.
The standard treatment for sarcoidosis involves corticosteroids such as prednisone. While steroid treatment may be effective, patients with sarcoidosis often have relapses unless they stay on steroids chronically. Unfortunately, long-term oral steroid use can cause substantial side effects including weight gain, osteoporosis, a depressed immune system, fluid retention, acne and thinning skin, and possibly diabetes. Therefore, researchers are interested in finding better, safer, longer-acting sarcoidosis treatments.
Since placenta-derived mesenchymal stem cells can modulate the immune system, researchers tested whether they could use these cells to improve symptoms of chronic lung sarcoidosis. Four patients with severe pulmonary sarcoidosis received intravenous infusions of 300 million placenta-derived mesenchymal stem cells. The cells were split into two doses, given one week apart. Patients underwent several tests to evaluate safety and efficacy of the stem cell treatment including X-rays, lung function testing, bronchoscopy, and bronchoalveolar lavage.
Stem cell treatment was associated with a temporary increase in pulmonary artery blood pressure. Two years after treatment, lung function was the same as it was at the beginning of the study. Impressively though, two patients were able to stop prednisone treatment and had visible improvements in their lungs on X-ray. One patient underwent prolonged remission (i.e., had no symptoms from sarcoidosis for an extended time period).
Because of their long-term complications, chronic steroid use is not an ideal treatment for any condition. With stem cell treatment, patients with chronic lung conditions could potentially decrease or minimize their daily intake of prednisone.

by admin | Jun 21, 2018 | Stem Cell Research
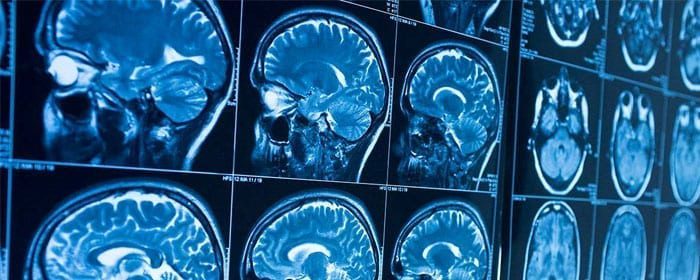
Traumatic brain injury is a potentially devastating condition that can that can often lead to permanent disability. While traumatic brain injury can be caused by a stroke, brain bleed, or head trauma, the resulting cascade of activity is similar regardless of the initial cause of the injury. Traumatic brain injury sets off a complex set of changes that causes brain cells to become dysfunctional and die. Consequently, people who suffer traumatic brain injury suffer cognitive, behavioral, and movement disorders depending on the area of the brain that is affected. Despite advances in neurocritical care, there are no effective treatments to reverse the cascade of traumatic brain injury.
Stem cells offer great promise in the treatment of traumatic brain injury. They work to change the harsh, destructive environment caused by this condition into one that can promote nerve cell protection and regeneration. To investigate stem cells as a potential defense against or treatment for traumatic brain injury, researchers have conducted several small clinical studies. Wang and colleagues performed a series of four stem cell transplantations over 5 to 7 days in patients that had suffered recent traumatic brain injury. No adverse effects were observed six months after treatment; however, patients did have a significant improvement in neurological function and a greater ability to care for themselves compared to brain-injured patients who did not receive stem cells. Chen and co-authors similarly showed that stem cells improved neurological function in patients who had suffered a recent stroke. As with Wang’s group, there were no observed side effects from treatment. Impressively, Chen’s group followed patients for up to 20 years after treatment and found no adverse events related to the stem cell treatment over that time.
As research continues, these initial results are particularly exciting because they are administered after—not before—brain injury. Based on these studies, stem cell treatment can be viewed as an intervention that can potentially be used to improve the outcome of traumatic brain injury it has occurred.

by admin | Jun 19, 2018 | Stem Cell Research
In a recent publication in the Journal of Clinical Oncology, Braiteh and colleagues describe the treatment of a 35-year-old white man who had suffered from psoriasis vulgaris and psoriatic arthropathy for 15 years before being diagnosed with multiple myeloma. Approximately 4 months after his initial treatment for the multiple myeloma, his disease relapsed. The patient was then treated with hematopoietic stem cells.
Before the stem cell treatment, the psoriasis plaques covered half of the patient’s body, with most of its coverage on the scalp, forehead, chest, abdomen, back, and ears. His elbows, knees, and nails were also affected. Following the treatment, the patient had a full year of remission of his myeloma and a complete regression of his psoriatic arthropathy and skin lesions. He was able to maintain this progress even without corticosteroids and phototherapy.
At the time of this publication, 15 months after his treatment, his myeloma had relapsed, but he was still psoriasis free and arthralgia free. These results demonstrate the possibility that hematopoietic stem cells could be an effective treatment option for patients suffering from psoriasis. Indeed, treatments with these types of stem cells have shown positive therapeutic effects in psoriasis and other autoimmune diseases, such as rheumatoid arthritis, multiple sclerosis, scleroderma, and systemic lupus erythematosus.
One reason it is thought that hematopoietic stem cells may help treat autoimmune disorders like psoriasis is that these cells may help the immune system to reduce its autoimmune activity. In autoimmune disorders, the immune system begins attacking one’s own body when it mistakes parts of the body as foreign and threatening. While this type of action by the immune system is favorable when the body has harmful foreign invaders like bacteria, it is problematic when the immune system mistakes innocuous stimuli as dangerous. Treatments that enable the immune system to recognize that non-threatening stimuli are safe or that they can reduce the negative reaction to the immune system as a result to these stimuli can be valuable when approaching autoimmune disorders.
With a better understanding of what stem cells can do to help these specific patient populations, it will be easier to develop treatment options that have a long term positive impact on individual patients.

by admin | Jun 11, 2018 | Stem Cell Research
Researchers have recently published their results from an experiment conducted to determine if a certain type of stem cell, called the umbilical cord-derived mesenchymal stem cell, has the potential for treating primary biliary cirrhosis. Primary biliary cirrhosis is a disease of the liver in which the bile ducts of the liver are increasingly destroyed over time.
In this disease, as the bile ducts are destroyed, bile and other toxins in the liver accumulate and lead to cholestasis. At this point, bile cannot flow out of the liver. Damage then occurs to the tissue of the liver, leading to scarring. As the scarring collects, excessive amounts of fibrous connective tissue form on the liver in what is called fibrosis. The late stage of fibrosis is known as cirrhosis, where the liver ceases to function properly.
The disease is autoimmune in nature, and because stem cells have shown promise for treating other types of autoimmune disease, presumably by impacting the activity of the immune system, researchers have reasoned that stem cells may also be beneficial for those suffering from primary biliary cirrhosis. In primary biliary cirrhosis, the body mistakes the liver, or aspects of it, as foreign and harmful and begins to attack it.
In their experiment, the scientists found that using umbilical cord-derived stem cells inhibited the problematic immune system activity associated with primary biliary cirrhosis. As a result, using umbilical cord-derived stem cells led to lower levels of problematic immune system components, as well as lower inflammation.
These results are a promising first step toward understanding how stem cells could potentially be used to treat primary biliary cirrhosis. As the research advances, it will become clearer how these cells can best be used to treat patients and how the cells have the impact they do on the liver and on disease progression.

by admin | Jun 8, 2018 | Stem Cell Research
Neurodegenerative conditions are neurological disorders that are caused by dysfunctions in part of the brain or nervous system which result in physical or psychological symptoms. They include conditions such as Alzheimer’s disease, Parkinson’s disease, ALS (Amyotrophic Lateral Sclerosis), and Multiple Sclerosis (MS), to name a few. In each of these conditions, certain areas of the brain or spinal cord become dysfunctional as nerve cells die. Stem cells have been considered as a potential source to help reverse or hold progression of symptoms. Research scientists have thoroughly reviewed the available scientific research on adipose (fat)-derived stem cells and have concluded that they show great promise in treating neurodegenerative conditions.
In an article from the Journal of Pharmacological Sciences, scientists explain that adipose-derived stem cells can play a unique and potentially beneficial role in many neurological conditions. For example, stem cells may be able to restore the learning and memory deficits that occur in Alzheimer’s disease by blocking or reducing the damage that occurs to the brain. Adipose-derived stem cells may be able to delay the progression of amyotrophic lateral sclerosis (ALS) or Lou Gehrig’s disease. In Huntington’s disease, a neurodegenerative condition that causes uncontrollable, dancelike movements, stem cells may be able to slow the rate at which nerve cells die in key areas of the brain. And for those with Parkinson’s disease, a neurological disease in which people have tremors, rigidity, and shuffling gait, stem cells show promise for relieving symptoms and slowing or halting the rate of progression.
The researchers also explored the possibility of adipose-derived stem cells treating other neurological conditions such as spinal cord injury and stroke. When people sustain damage to the spinal cord, one of the reasons that they cannot regain function is due to scar tissue forming at the site of injury. Adipose-derived stem cells may be able to block these scars, and potentially help nerve cell fibers regrow. During a stroke, there is a lack of blood flow to the brain causing blood cells to die. Adipose-derived stem cells may be able to reduce the amount of damage to brain tissue and allow patients to preserve more of their natural abilities.
In all of these cases, continued clinical trials are needed to demonstrate the positive effect of adipose-derived stem cells. Nonetheless, the scientific evidence is strong in supporting these treatments. As the authors of the review state, adipose-derived stem cells are “very likely to be a promising clinical option for the treatment of diverse neurological disorder patients.”

by admin | Jun 4, 2018 | Stem Cell Research
In order to tell other parts of the body how to react, cells must be able to communicate. For many years, scientists have known that cells do this through chemical secretion, which releases a message to other cells. It wasn’t until recently, however, that researchers discovered the ability to deliver these messages through extracellular vesicles.
In some cases, these extracellular vesicles bud directly from a cell membrane. In other instances, tinier vesicles are assembled inside the cell before being released through the membrane. These are called exosomes, and they carry sophisticated RNA.
What Do Exosomes Do?
Recent studies tell us that exosomes play a critical role in cell-to-cell communication. Exosomes formed by specific cells can perform multiple functions, including exerting positive effects on tissue regeneration. Yet, not all exosomes are good: viruses, too, rely on these cellular pathways to communicate and replicate. In other words, infected cells also use exosomes to progress diseases.
Exosomes & Their Potential Power
Most recently, research has confirmed that cellular communication via exosomes is the process through which infectious diseases like malaria progress. Traditionally, high levels of viruses or bacteria in the blood were needed for accurate disease detection. Now, however, researchers believe that monitoring changes in exosomes – which are always present in human plasma – could aid in the ability to diagnose submicroscopic infections.
Yet, the potential impact of exosomes on modern medicine extends far beyond diagnostics. Secreted exosomes derived from stem cells could hold serious therapeutic potential for many conditions. Using stem cells as a therapy for conditions such as liver disease, for instance, shows significant potential. Stem cell-secreted exosomes present a cell-free form of therapy.
Unlike their parent cells, the exosomes are smaller, less complex, and easier to produce and store. Researchers are also exploring the potential of stem cell-secreted exosomes for treating conditions like autoimmune uveitis, an inflammatory eye condition. Additionally, the process is being looked into as a cell-free therapy for organ repair. In conditions such as cardiovascular disease, damaged tissue is unable to repair itself. Exosomes derived from stem cells could aid in myocardial repair, according to recent research. They have also been recently implemented as a way to track the progression of spinal muscular atrophy (SMA), including the way its patients respond to treatments.
Because exosome studies are still being performed, the full impact these powerful vesicles will have on medicine is still unknown. However, based on the sheer volume of exosome research currently being conducted, it’s clear that scientists believe they could hold the answers to some of our most pressing medical questions.

 St. Petersburg, Florida
St. Petersburg, Florida
